( uncountable ) The tendency of a system that is left to itself to descend into chaos. 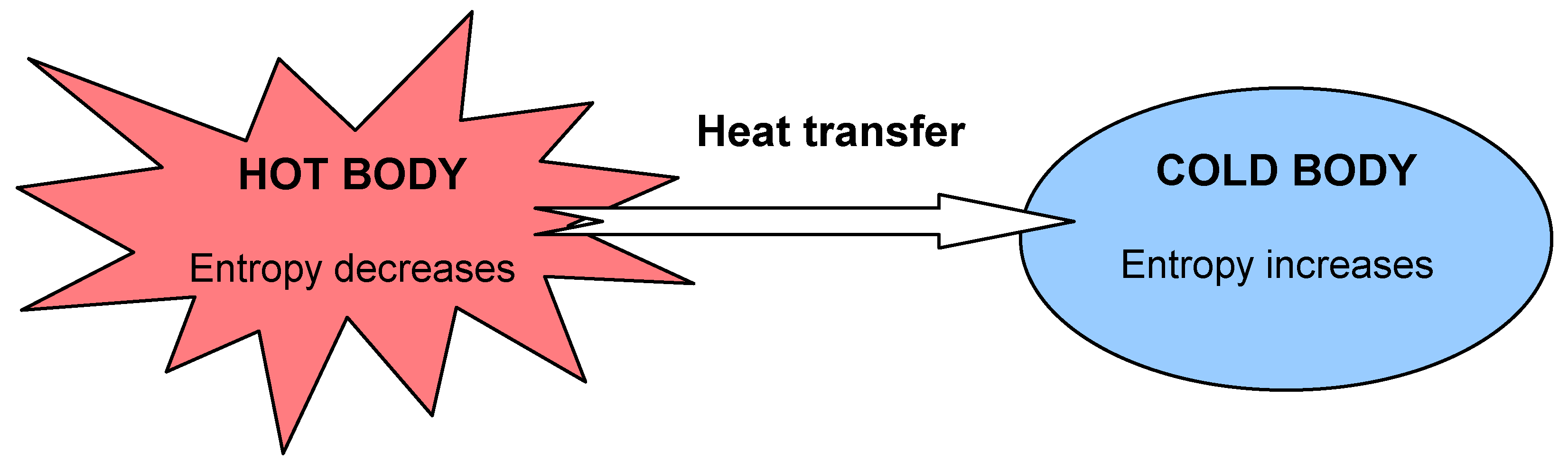
( statistics, information theory, countable ) A measure of the amount of information and noise present in a signal.The dispersal of energy how much energy is spread out in a process, or how widely spread out it becomes, at a specific temperature.The capacity factor for thermal energy that is hidden with respect to temperature.( thermodynamics, countable ) A measure of the amount of energy in a physical system that cannot be used to do work.( Boltzmann definition ) A measure of the disorder directly proportional to the natural logarithm of the number of microstates yielding an equivalent thermodynamic macrostate.A measure of the disorder present in a system.Where n and m are the coefficients found in the balanced chemical equation of the reaction.Entropy ( countable and uncountable, plural entropies) Entropy is a fundamental concept in Data Science because it shows up all over the place - from Decision Trees, to similarity metrics, to state of the art dim. The entropy change for imaginary reversible process is same as that of an irreversible process between given final and initial states. The entropy change of a reaction where the reactants and products are in their standard state can be determined using the following equation: (Source: UC Davis ChemWiki by University of California\CC-BY-SA-3.0) Standard Entropy Change of a Reaction, Δ S° According to the Boltzmann equation, entropy is a measure of the number of microstates available to a system. Temperature of a Single Substance.” This is a generalized plot of entropy versus temperature for a single substance. These large increases occur due to sudden increased molecular mobility and larger available volumes associated with the phase changes.įigure 18.3 “Entropy vs. This can be seen in Figure 18.3 “Entropy vs. Temperature of a Single Substance.” Large jumps in entropy occur at the phase changes: solid to liquid and liquid to gas. Entropy is a powerful tool for quantification of the brain function and its information processing capacity. The standard molar entropy of any substance increases as the temperature increases.Gases tend to have much larger standard molar enthalpies than liquids, and liquids tend to have larger values than solids, when comparing the same or similar substances. Entropy concept is frequently used in many scientific disciplines: physics 1-70, equilibrium and non- equilibrium thermodynamics 1-25, 60-70, statistical.There are more possible arrangements of atoms in space for larger, more complex molecules, increasing the number of possible microstates. Larger, more complex molecules have higher standard molar enthalpy values than smaller or simpler molecules.Look up any word in the dictionary offline, anytime. The more disordered a system is, the higher (the more positive) the value of. entropy (specialist) a way of measuring the lack of order that exists in a system. This definition, of thermodynamic entropy, depends upon cyclic processes that can reach the equality, which are called reversible processes. Several trends emerge from standard molar entropy data: Entropy can be defined as the randomness or dispersal of energy of a system. An adiabatic process (one which does not generate any heat) from state A to state B is therefore only possible if it is entropy increasing: S (A) S (B). Table 18.1c Standard Molar Entropies of Selected Solids at 298 K Solid Table 18.1b Standard Molar Entropies of Selected Liquids at 298 K Liquid Table 18.1a Standard Molar Entropies of Selected Gases at 298 K Gas These values have been tabulated, and selected substances are listed in Table 18.1a to c “Standard Molar Entropies of Selected Substances at 298 K”. One equation is Boltzmann’s equation: S kln (W), where S is entropy (the usual variable for entropy), k is Boltzmann’s constant which is equal to the gas constant divided by Avogadro’s number which is approximately equal to 1.38 x 10 (-23) J/K, and W is the number of microstates which is a unitless quantity. The standard molar entropy, S°, is the entropy of 1 mole of a substance in its standard state, at 1 atm of pressure. Why Does Entropy Matter for Your Life Heres the. Assume the change is reversible and the temperature remains constant. And there are always far more disorderly variations than orderly ones. Determine the change in entropy (in J/K) of water when 425 kJ of heat is applied to it at 50☌.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
